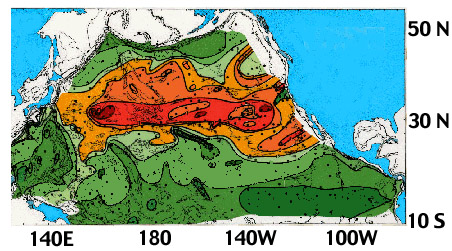
Figure MS-1. Quartz concentration in North Pacific sediments (carbonate-free basis) showing effects of eolian transport in westerlies and northeast trades.
Marine sediments are the products of a limited number of physical, biological, and chemical processes. The nature of the resultant sediments is determined by the relative rates of input of material supplied by these processes. In this class we focus on deep sea sediments, but the processes apply equally to shallow water deposits.
Physical processes dominate at ocean margins, where they transfer particles eroded from the land to the sea floor.
Active sedimentation processes (where the sediment modifies the properties and behavior of the suspension) include mass wasting and density currents. Such deposition tends to mask other sedimentary processes. The giant landslides surrounding the Hawaiian islands and the abyssal plains of the Atlantic are striking deep-sea examples of active sedimentation. Because active sedimentation depends on gravitational energy, it does not extend seaward of the trenches along convergent plate boundaries (but can affect the entire ocean basin off passive margins).
Passive sedimentationprocesses are those in which the sediment is carried by but does not modify the normal thermohaline circulation. Examples:

Sediments formed by physical processes have distinctive acoustic signatures of military interest. Hence they have been much studied during the past 50 years.
"Passive" sediments record the history of deep currents, volcanism, aridity, wind trajectories, and iceberg abundances and trajectories. Drift deposits, which can accumulate at hundreds of meters per million years, yield some of the highest resolution paleoceanographic records.
Biological processes dominate sediment formation in areas of high productivity that receive little terrigenous material. The equatorial Pacific and Southern Ocean are examples.
Several taxa of phytoplankton and zooplankton (the latter include benthic and upper water column representatives) secrete either CaCO3 (coccolithophores and foraminiferans) or opal (hydrated SiO2 - diatoms and radiolarians). If not masked by terrigenous material, the tests (shells) of these organisms can form carbonate or siliceous oozes.
Because the distribution and abundance of the various species are determined by the temperature, salinity, thermocline depth, carbonate chemistry, and productivity of the waters in which they live, most of what we know about paleoceanography is derived from fossil assemblages of these organisms.
In addition, the composition of the tests records the oxygen and carbon isotopic compositions and trace element contents of the waters in which they were secreted. These parameters in fossil tests provide insights to past ice volumes, temperature, productivity, and changes in biogeochemical cycles.
Benthic organisms also modify the historical record by actively mixing ("bioturbating") the most recently deposited sediments. Bioturbation, which is addressed later in the course, is effectively a low-pass filter that supresses or eliminates records of events that create layers of sediment thinner than the depth of mixing. Rapidly deposited or anoxic sediments provide the only deep-sea records capable of resolving events shorter than about a millennium.
Chemical processes dominate sedimentation only in deep, low productivity areas shielded from terrigenous material. Precipitates from hydrothermal solutions emanating from mid-ocean ridges are prominent along the flanks of the East Pacific Rise in the South Pacific. Authigenic deposits (formed by the very slow precipitation of oxyhydroxides and silicates? from normal seawater) form distinctive sediments in the central South Pacific as well as ferromanganese nodules (Figure MS-2) and crusts on any surfaces where other sediments are absent or accumulating extremely slowly.
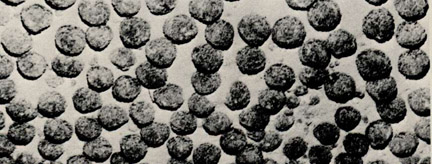
Figure MS-2. Deep sea ferromanganese nodules on the floor of the South Pacific Ocean (individual nodules are 5-10 cm diameter).
Chemical processes also modify biological (biogenic) sediments through the dissolution of CaCO3 and (to a lesser extent) opal in deep water. T and P both play a role in increasing the corrosiveness of deep waters, but the major effect on carbonate tests is due to the creation of CO2 by the biologically mediated oxidation of organic matter, which creates biocarbonate ions at the expense of carbonate ions (Equn. MS-1), thereby driving the dissolution of carbonate tests.
Equn. MS-1. CO2 + CO3= + H2O = 2HCO3-
In general, the carbonate content of deep sea sediments decreases with increasing water depth. Two important horizons are the lysocline, where the proportion of solution-resistant tests increases abruptly, and the calcite compensation depth (CCD) which is the boundary between carbonate-bearing and carbonate-free sediments (Figure MS-3)
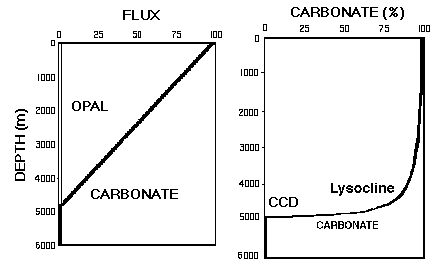
Figure MS-3. Carbonate concentration versus depth showing lysocline and CCD (right) created by simple linear dissolution with depth of carbonate in a carbonate-opal mixture (left).
The sea floor distributions of both carbonate and opal tests reflect the evolution of bottom water chemistry as it makes the "grand tour" from the North Atlantic via the Southern Ocean to the northern Pacific and Indian Oceans. As the bottom water "ages" it accumulates carbon dioxide, which drives equation MS-1 to the right, thereby dissolving CaCO3.
Chemical reactions within the sea floor (diagenesis) can further modify deep-sea sediments, both through isochemical changes (which can obliterate particle source and paleomagnetic information) and open-system changes (which can modify the isotopic and trace element composition of sedimentary particles).
Rates of deposition. Sediments can be dated by a number of well established techniques. the most common are:
From dated layers in sediment cores, the rates of sedimentation of the various types of deposits can be calculated. Typical ranges for the products of the various depositional processes are (note that m/my is equivalent to mm/ky):
|
Nearshore sediments, turbidites |
Up to km/my (kilometers/million years) |
|
Hemipelagic deposits |
Tens to hundreds of m/my |
|
Drift deposits |
40-400 m/my |
|
Mid-latitude eolian deposits |
3 to 10 m/my |
|
Ice rafted material |
10+ m/my |
|
Carbonate oozes |
Up to 50 m/my |
|
Siliceous oozes |
Up to 10 m/my |
|
Hydrothermal deposits (off ridge axes) |
About 0.5 m/my |
|
Hydrogenous sediments |
Rarely exceed 0.2 m/my |
|
Ferromanganese nodules |
0.0002 to 0.005 m/my (0.2 to 5 mm/my) |
|
Oceanography 540 Pages |